SepMate™-15 (IVD)
Tube for density gradient centrifugation for in vitro diagnostic (IVD) applications
概要
SepMate™ is now manufactured under cGMP and registered as an In Vitro Diagnostic (IVD) device in Australia, Canada, Europe and USA only. In China SepMate™ is considered a non-medical device by the China Food and Drug Administration (CFDA), and should therefore be used as general laboratory equipment. The end user is responsible for determining whether the product is suitable for their specific application. Due to these changes, users in Australia, Canada, Europe, USA and China should, from now on, order SepMate™-15 (IVD) (Catalog #85415 and #85420) and SepMate™-50 (IVD) (Catalog #85450 and #85460). Users in all other countries please refer to SepMate™-15 (RUO) (Catalog #86415) and SepMate™-50 (RUO) (Catalog #86450). Changes to SepMate™ have no impact on the product composition, performance or protocol.
SepMate™ is a tube that facilitates the isolation of PBMCs or specific cell types by density gradient centrifugation. SepMate™ tubes contain an insert that provides a barrier between the density gradient medium and blood. SepMate™ eliminates the need for careful layering of blood onto the density gradient medium, and allows for fast and easy harvesting of the isolated mononuclear cells with a simple pour. SepMate™ is also compatible with RosetteSep™ enrichment cocktails, allowing isolation of specific cell types in less than 30 minutes.
SepMate™ is a tube that facilitates the isolation of PBMCs or specific cell types by density gradient centrifugation. SepMate™ tubes contain an insert that provides a barrier between the density gradient medium and blood. SepMate™ eliminates the need for careful layering of blood onto the density gradient medium, and allows for fast and easy harvesting of the isolated mononuclear cells with a simple pour. SepMate™ is also compatible with RosetteSep™ enrichment cocktails, allowing isolation of specific cell types in less than 30 minutes.
Advantages
• Eliminates the need for carefully layering blood over the density gradient medium (e.g. Lymphoprep™, etc.)
• Reduces total centrifuge time to 10 minutes with the brake on for fresh samples
• Allows fast and easy harvesting of the isolated mononuclear cells by simply pouring off the supernatant
• Can be combined with RosetteSep™ enrichment cocktails to isolate specific cell types in just 30 minutes
• Reduces total centrifuge time to 10 minutes with the brake on for fresh samples
• Allows fast and easy harvesting of the isolated mononuclear cells by simply pouring off the supernatant
• Can be combined with RosetteSep™ enrichment cocktails to isolate specific cell types in just 30 minutes
Components
- SepMate™-15 (IVD), 100 Tubes (Catalog #85415)
- Dispenser box containing 4 bags, 25 Tubes/Bag
- SepMate™-15 (IVD), 500 Tubes (Catalog #85420)
- Dispenser box containing 4 bags, 25 Tubes/Bag (Catalog #85415) x 5
Contains
Polypropylene tube containing an insert
Subtype
Centrifugation Tubes
Cell Type
B Cells, Dendritic Cells, Monocytes, Mononuclear Cells, NK Cells, T Cells, T Cells, CD4+, T Cells, CD8+, T Cells, Other Subsets, T Cells, Regulatory
Species
Human
Sample Source
Bone Marrow, Cord Blood, Whole Blood
Selection Method
Negative
Application
Cell Isolation, In Vitro Diagnostic
Brand
SepMate
Area of Interest
Chimerism, HLA, Immunology
技术资料
| Document Type | 产品名称 | Catalog # | Lot # | 语言 |
|---|---|---|---|---|
| Product Information Sheet | SepMate™-15 (IVD) | 85415, 85420 | All | English |
数据及文献
Data
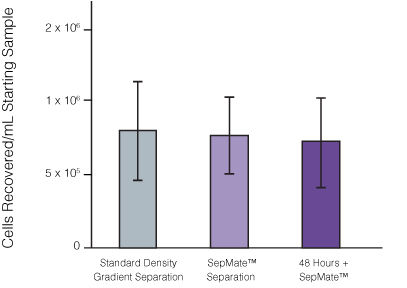
Figure 1. Recovery of mononuclear cells (MNCs) from peripheral blood using SepMate™-50 versus standard density gradient centrifiguation.
Recovery of MNCs from fresh and 48-hour post blood draw enriched by density gradient centrifugation with SepMate™ (purple) or without (grey). There was no significant difference in the recovery of MNCS with and without SepMate™.
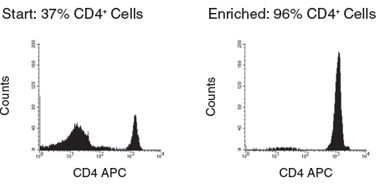
Figure 2. Human CD4+ T Cell Isolation using SepMate™-50 and RosetteSep™ Human CD4+ T Cell Enrichment Cocktail
Publications (30)
Science advances 2020 jun
A highly potent lymphatic system-targeting nanoparticle cyclosporine prevents glomerulonephritis in mouse model of lupus.
Abstract
Abstract
Cyclosporine A (CsA) is a powerful immunosuppressant, but it is an ineffective stand-alone treatment for systemic lupus erythematosus (SLE) due to poor target tissue distribution and renal toxicity. We hypothesized that CD71 (transferrin receptor 1)-directed delivery of CsA to the lymphatic system would improve SLE outcomes in a murine model. We synthesized biodegradable, ligand-conjugated nanoparticles [P2Ns-gambogic acid (GA)] targeting CD71. GA conjugation substantially increased nanoparticle association with CD3+ or CD20+ lymphocytes and with intestinal lymphoid tissues. In orally dosed MRL-lpr mice, P2Ns-GA-encapsulated CsA increased lymphatic drug delivery 4- to 18-fold over the ligand-free formulation and a commercial CsA capsule, respectively. Improved lymphatic bioavailability of CsA was paralleled by normalization of anti-double-stranded DNA immunoglobulin G titer, plasma cytokines, and glomerulonephritis. Thus, this study demonstrates the translational potential of nanoparticles that enhance the targeting of lymphatic tissues, transforming CsA into a potent single therapeutic for SLE.
Nature 2020
A dominant autoinflammatory disease caused by non-cleavable variants of RIPK1
Abstract
Abstract
Activation of RIPK1 controls TNF-mediated apoptosis, necroptosis and inflammatory pathways1. Cleavage of human and mouse RIPK1 after residues D324 and D325, respectively, by caspase-8 separates the RIPK1 kinase domain from the intermediate and death domains. The D325A mutation in mouse RIPK1 leads to embryonic lethality during mouse development2,3. However, the functional importance of blocking caspase-8-mediated cleavage of RIPK1 on RIPK1 activation in humans is unknown. Here we identify two families with variants in RIPK1 (D324V and D324H) that lead to distinct symptoms of recurrent fevers and lymphadenopathy in an autosomal-dominant manner. Impaired cleavage of RIPK1 D324 variants by caspase-8 sensitized patients' peripheral blood mononuclear cells to RIPK1 activation, apoptosis and necroptosis induced by TNF. The patients showed strong RIPK1-dependent activation of inflammatory signalling pathways and overproduction of inflammatory cytokines and chemokines compared with unaffected controls. Furthermore, we show that expression of the RIPK1 mutants D325V or D325H in mouse embryonic fibroblasts confers not only increased sensitivity to RIPK1 activation-mediated apoptosis and necroptosis, but also induction of pro-inflammatory cytokines such as IL-6 and TNF. By contrast, patient-derived fibroblasts showed reduced expression of RIPK1 and downregulated production of reactive oxygen species, resulting in resistance to necroptosis and ferroptosis. Together, these data suggest that human non-cleavable RIPK1 variants promote activation of RIPK1, and lead to an autoinflammatory disease characterized by hypersensitivity to apoptosis and necroptosis and increased inflammatory response in peripheral blood mononuclear cells, as well as a compensatory mechanism to protect against several pro-death stimuli in fibroblasts.
Frontiers in immunology 2020
Rheumatoid Arthritis Patients, Both Newly Diagnosed and Methotrexate Treated, Show More DNA Methylation Differences in CD4+ Memory Than in CD4+ Na\ive T Cells."
Abstract
Abstract
Background: Differences in DNA methylation have been reported in B and T lymphocyte populations, including CD4+ T cells, isolated from rheumatoid arthritis (RA) patients when compared to healthy controls. CD4+ T cells are a heterogeneous cell type with subpopulations displaying distinct DNA methylation patterns. In this study, we investigated DNA methylation using reduced representation bisulfite sequencing in two CD4+ T cell populations (CD4+ memory and na{\{i}}ve cells) in three groups: newly diagnosed disease modifying antirheumatic drugs (DMARD) na{\"{i}}ve RA patients (N = 11) methotrexate (MTX) treated RA patients (N = 18) and healthy controls (N = 9) matched for age gender and smoking status. Results: Analyses of these data revealed significantly more differentially methylated positions (DMPs) in CD4+ memory than in CD4+ na{\""{i}}ve T cells (904 vs. 19 DMPs) in RA patients compared to controls. The majority of DMPs (72{\%}) identified in newly diagnosed and DMARD na{\""{i}}ve RA patients with active disease showed increased DNA methylation (39 DMPs) whereas most DMPs (80{\%}) identified in the MTX treated RA patients in remission displayed decreased DNA methylation (694 DMPs). Interestingly we also found that about one third of the 101 known RA risk loci overlapped (±500 kb) with the DMPs. Notably introns of the UBASH3A gene harbor both the lead RA risk SNP and two DMPs in CD4+ memory T cells. Conclusion: Our results suggest that RA associated DNA methylation differences vary between the two T cell subsets but are also influenced by RA characteristics such as disease activity disease duration and/or MTX treatment."""
Science immunology 2020
Longitudinal immune profiling reveals key myeloid signatures associated with COVID-19.
Abstract
Abstract
COVID-19 pathogenesis is associated with an exaggerated immune response. However, the specific cellular mediators and inflammatory components driving diverse clinical disease outcomes remain poorly understood. We undertook longitudinal immune profiling on both whole blood and peripheral blood mononuclear cells (PBMCs) of hospitalized patients during the peak of the COVID-19 pandemic in the UK. Here, we report key immune signatures present shortly after hospital admission that were associated with the severity of COVID-19. Immune signatures were related to shifts in neutrophil to T cell ratio, elevated serum IL-6, MCP-1 and IP-10, and most strikingly, modulation of CD14+ monocyte phenotype and function. Modified features of CD14+ monocytes included poor induction of the prostaglandin-producing enzyme, COX-2, as well as enhanced expression of the cell cycle marker Ki-67. Longitudinal analysis revealed reversion of some immune features back to the healthy median level in patients with a good eventual outcome. These findings identify previously unappreciated alterations in the innate immune compartment of COVID-19 patients and lend support to the idea that therapeutic strategies targeting release of myeloid cells from bone marrow should be considered in this disease. Moreover, they demonstrate that features of an exaggerated immune response are present early after hospital admission suggesting immune-modulating therapies would be most beneficial at early timepoints.
Frontiers in immunology 2020
Single-Cell Atlas Reveals Complexity of the Immunosuppressive Microenvironment of Initial and Recurrent Glioblastoma.
Abstract
Abstract
The Glioblastoma (GBM) immune microenvironment plays a critical role in tumor development, progression, and prognosis. A comprehensive understanding of the intricate milieu and its interactions remains unclear, and single-cell analysis is crucially needed. Leveraging mass cytometry (CyTOF), we analyzed immunocytes from 13 initial and three recurrent GBM samples and their matched peripheral blood mononuclear cells (pPBMCs). Using a panel of 30 markers, we provide a high-dimensional view of the complex GBM immune microenvironment. Hematoxylin and eosin staining and polychromatic immunofluorescence were used for verification of the key findings. In the initial and recurrent GBMs, glioma-associated microglia/macrophages (GAMs) constituted 59.05 and 27.87{\%} of the immunocytes, respectively; programmed cell death-ligand 1 (PD-L1), T cell immunoglobulin domain and mucin domain-3 (TIM-3), lymphocyte activation gene-3 (LAG-3), interleukin-10 (IL-10) and transforming growth factor-$\beta$ (TGF$\beta$) demonstrated different expression levels in the GAMs among the patients. GAMs could be subdivided into different subgroups with different phenotypes. Both the exhausted T cell and regulatory T (Treg) cell percentages were significantly higher in tumors than in pPBMCs. The natural killer (NK) cells that infiltrated into the tumor lesions expressed higher levels of CXC chemokine receptor 3 (CXCR3), as these cells expressed lower levels of interferon-$\gamma$ (IFN$\gamma$). The immune microenvironment in the initial and recurrent GBMs displayed similar suppressive changes. Our study confirmed that GAMs, as the dominant infiltrating immunocytes, present great inter- and intra-tumoral heterogeneity and that GAMs, increased exhausted T cells, infiltrating Tregs, and nonfunctional NK cells contribute to local immune suppressive characteristics. Recurrent GBMs share similar immune signatures with the initial GBMs except the proportion of GAMs decreases.
European journal of human genetics : EJHG 2019 may
Biallelic variants in POLR3GL cause endosteal hyperostosis and oligodontia.
Abstract
Abstract
RNA polymerase III (Pol III) is an essential 17-subunit complex responsible for the transcription of small housekeeping RNAs such as transfer RNAs and 5S ribosomal RNA. Biallelic variants in four genes (POLR3A, POLR3B, and POLR1C and POLR3K) encoding Pol III subunits have previously been found in individuals with (neuro-) developmental disorders. In this report, we describe three individuals with biallelic variants in POLR3GL, a gene encoding a Pol III subunit that has not been associated with disease before. Using whole exome sequencing in a monozygotic twin and an unrelated individual, we detected homozygous and compound heterozygous POLR3GL splice acceptor site variants. RNA sequencing confirmed the loss of full-length POLR3GL RNA transcripts in blood samples of the individuals. The phenotypes of the described individuals are mainly characterized by axial endosteal hyperostosis, oligodontia, short stature, and mild facial dysmorphisms. These features largely fit within the spectrum of phenotypes caused by previously described biallelic variants in POLR3A, POLR3B, POLR1C, and POLR3K. These findings further expand the spectrum of POLR3-related disorders and implicate that POLR3GL should be included in genetic testing if such disorders are suspected.