MethoCult™ H4531
Methylcellulose-based medium with agar-LCM (without erythropoietin [EPO]) for human cells
概要
MethoCult™ H4531 is methylcellulose-based medium for the growth and enumeration of hematopoietic progenitor cells in colony-forming unit (CFU) assays of human bone marrow, peripheral blood, mobilized peripheral blood, and cord blood samples. This formulation contains agar leukocyte conditioned medium (agar-LCM) as a source of colony-stimulating factors. MethoCult™ H4531 supports the growth of granulocyte-macrophage progenitor cells (CFU-GM, CFU-G and CFU-M) and EPO-independent erythroid progenitor cells. It is suitable for detection of EPO-independent erythroid progenitor cells.
Browse our Frequently Asked Questions (FAQs) on performing the CFU assay and explore its utility as part of the cell therapy workflow.
Browse our Frequently Asked Questions (FAQs) on performing the CFU assay and explore its utility as part of the cell therapy workflow.
Contains
• Methylcellulose in Iscove's MDM
• Fetal bovine serum
• Bovine serum albumin
• 2-Mercaptoethanol
• Agar-LCM
• Supplements
• Fetal bovine serum
• Bovine serum albumin
• 2-Mercaptoethanol
• Agar-LCM
• Supplements
Subtype
Semi-Solid Media, Specialized Media
Cell Type
Hematopoietic Stem and Progenitor Cells
Species
Human
Application
Cell Culture, Colony Assay, Functional Assay
Brand
MethoCult
Area of Interest
Stem Cell Biology
技术资料
| Document Type | 产品名称 | Catalog # | Lot # | 语言 |
|---|---|---|---|---|
| Product Information Sheet | MethoCult™ H4531 | 04531 | All | English |
| Manual | MethoCult™ H4531 | 04531 | All | English |
| Safety Data Sheet | MethoCult™ H4531 | 04531 | All | English |
数据及文献
Data
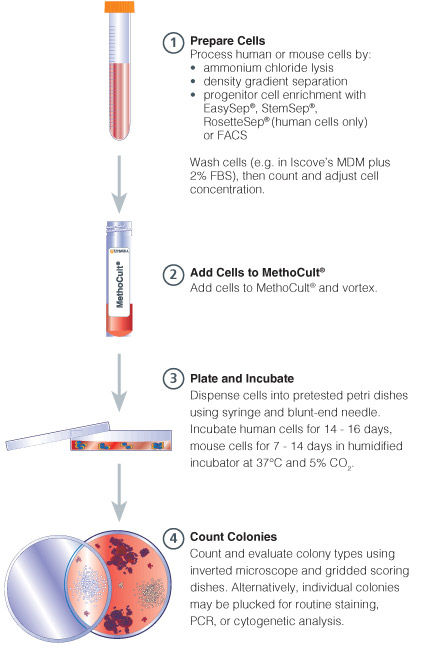
Figure 1. Procedure Summary for Hematopoietic CFU Assays
Publications (11)
Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association 2012 FEB
mTOR inhibition and erythropoiesis: microcytosis or anaemia?
Abstract
Abstract
BACKGROUND: Anaemia and microcytosis are common post kidney transplantation. The aim of this study was to evaluate the potential role of mammalian target of rapamycin (mTOR) inhibition in the development of anaemia and microcytosis in healthy animals and in human erythroid cultures in vitro. METHODS: Rats with normal kidney function were treated with sirolimus (n = 7) or vehicle (n = 8) for 15 weeks. Hemograms were determined thereafter. In the sirolimus withdrawal part of the study, rats received sirolimus (SRL) for 67 days (n = 4) 1 mg/kg three times per week or for 30 days (n = 4) and were observed until Day 120. Hemograms were performed regularly. Peripheral blood mononuclear cells from healthy controls (HC; n = 8), kidney transplant patients with sirolimus treatment with (SRL + MC; n = 8) or without microcytosis (SRL - MC; n = 8) were isolated and cultured in the absence or presence of SRL (5 ng/mL). RESULTS: SRL-treated animals had a reduced mean corpuscular volume (MCV) and elevated erythrocyte count compared with control animals after 15 weeks of treatment. This effect was evident as early as 4 weeks (MCV: 61.5 ± 1.8 versus 57 ± 1.7 fL; P = 0.0156; Red blood count 7.4 ± 0.3 × 10(9)/L versus 8.6 ± 0.5 × 10(9)/L; P = 0.0156) and was reversible 90 days after SRL withdrawal. SRL in the culture medium of erythroid cultures led to fewer colonies in cultures from HC as well as from kidney transplant patients (without SRL: 34.2 ± 11.4 versus with SRL: 27.5 ± 9.9 BFU-E-derived colonies P = 0.03), regardless if the cultures were derived from recipients with normocytic or with microcytic erythrocytes. The presence of tacrolimus in the culture medium had no influence on the number and size of colonies. CONCLUSION: mTOR inhibition induces microcytosis and polyglobulia, but not anaemia in healthy rats. This might be caused by growth inhibition of erythroid precursor cells.
Journal of medical genetics 2010 OCT
Germline mutations of the CBL gene define a new genetic syndrome with predisposition to juvenile myelomonocytic leukaemia.
Abstract
Abstract
BACKGROUND: CBL missense mutations have recently been associated with juvenile myelomonocytic leukaemia (JMML), an aggressive myeloproliferative and myelodysplastic neoplasm of early childhood characterised by excessive macrophage/monocyte proliferation. CBL, an E3 ubiquitin ligase and a multi-adaptor protein, controls proliferative signalling networks by downregulating the growth factor receptor signalling cascades in various cell types. METHODS AND RESULTS: CBL mutations were screened in 65 patients with JMML. A homozygous mutation of CBL was found in leukaemic cells of 4/65 (6%) patients. In all cases, copy neutral loss of heterozygosity of the 11q23 chromosomal region, encompassing the CBL locus, was demonstrated. Three of these four patients displayed additional features suggestive of an underlying developmental condition. A heterozygous germline CBL p.Y371H substitution was found in each of them and was inherited from the father in one patient. The germline mutation represents the first hit, with somatic loss of heterozygosity being the second hit positively selected in JMML cells. The three patients display a variable combination of dysmorphic features, hyperpigmented skin lesions and microcephaly that enable a 'CBL syndrome' to be tentatively delineated. Learning difficulties and postnatal growth retardation may be part of the phenotype. CONCLUSION: A report of germline mutations of CBL in three patients with JMML is presented here, confirming the existence of an unreported inheritable condition associated with a predisposition to JMML.
Proceedings of the National Academy of Sciences of the United States of America 2010 FEB
Construction and genetic selection of small transmembrane proteins that activate the human erythropoietin receptor.
Abstract
Abstract
This work describes a genetic approach to isolate small, artificial transmembrane (TM) proteins with biological activity. The bovine papillomavirus E5 protein is a dimeric, 44-amino acid TM protein that transforms cells by specifically binding and activating the platelet-derived growth factor beta receptor (PDGFbetaR). We used the E5 protein as a scaffold to construct a retrovirus library expressing approximately 500,000 unique 44-amino acid proteins with randomized TM domains. We screened this library to select small, dimeric TM proteins that were structurally unrelated to erythropoietin (EPO), but specifically activated the human EPO receptor (hEPOR). These proteins did not activate the murine EPOR or the PDGFbetaR. Genetic studies with one of these activators suggested that it interacted with the TM domain of the hEPOR. Furthermore, this TM activator supported erythroid differentiation of primary human hematopoietic progenitor cells in vitro in the absence of EPO. Thus, we have changed the specificity of a protein so that it no longer recognizes its natural target but, instead, modulates an entirely different protein. This represents a novel strategy to isolate small artificial proteins that affect diverse membrane proteins. We suggest the word traptamer" for these transmembrane aptamers."
Cancer research 2003 NOV
In vitro tests of the validity of singlet oxygen luminescence measurements as a dose metric in photodynamic therapy.
Abstract
Abstract
Singlet oxygen ((1)O(2)) is widely believed to be the major cytotoxic agent involved in photodynamic therapy (PDT). We showed recently that measurement of the weak near infrared luminescence of (1)O(2) is possible in cells in vitro and tissues in vivo. Here, we investigated the relationship between the integrated luminescence signal and the in vitro PDT response of AML5 leukemia cells sensitized with aminolevulinic acid-induced protoporphyrin IX (PpIX). Sensitized cell suspensions were irradiated with pulsed 523 nm laser light at average fluence rates of 10, 25, or 50 mWcm(-2) and, (1)O(2) luminescence measurements were made throughout the treatment. Cell survival was measured with either propidium iodide-labeled flow cytometry or colony-forming assay. The PpIX concentration in the cells, the photobleaching, and the pO(2) in the cell suspensions were also monitored. There were large variations in cell survival and (1)O(2) generation in different experiments due to different controlled treatment parameters (fluence and fluence rate) and other uncontrolled factors (PpIX synthesis and oxygenation). However, in all of the cases, cell kill correlated strongly with the cumulative (1)O(2) luminescence and allowed direct estimation of the (1)O(2) per cell required to achieve a specific level of cell kill. This study supports the validity and potential utility of (1)O(2) luminescence measurement as a dosimetric tool for PDT, as well as confirming the likely role of (1)O(2) in porphyrin-based PDT.
Blood 2003 APR
Discrimination of polycythemias and thrombocytoses by novel, simple, accurate clonality assays and comparison with PRV-1 expression and BFU-E response to erythropoietin.
Abstract
Abstract
Essential thrombocythemia (ET) and polycythemia vera (PV) are clonal myeloproliferative disorders that are often difficult to distinguish from other causes of elevated blood cell counts. Assays that could reliably detect clonal hematopoiesis would therefore be extremely valuable for diagnosis. We previously reported 3 X-chromosome transcription-based clonality assays (TCAs) involving the G6PD, IDS, and MPP1 genes, which together were informative in about 65% of female subjects. To increase our ability to detect clonality, we developed simple TCA for detecting the transcripts of 2 additional X-chromosome genes: Bruton tyrosine kinase (BTK) and 4-and-a-half LIM domain 1 (FHL1). The combination of TCA established the presence or absence of clonal hematopoiesis in about 90% of female subjects. We show that both genes are subject to X-chromosome inactivation and are polymorphic in all major US ethnic groups. The 5 TCAs were used to examine clonality in 46 female patients along with assays for erythropoietin-independent erythroid colonies (EECs) and granulocyte PRV-1 mRNA levels to discriminate polycythemias and thrombocytoses. Of these, all 19 patients with familial polycythemia or thrombocytosis had polyclonal hematopoiesis, whereas 22 of 26 patients with clinical evidence of myeloproliferative disorder and 1 patient with clinically obscure polycythemia were clonal. Interestingly, interferon alpha therapy in 2 patients with PV was associated with reversion of clonal to polyclonal hematopoiesis. EECs were observed in 14 of 14 patients with PV and 4 of 12 with ET, and increased granulocyte PRV-1 mRNA levels were found in 9 of 13 patients with PV and 2 of 12 with ET. Thus, these novel clonality assays are useful in the diagnosis and follow-up of polycythemic conditions and disorders with increased platelet levels.
Blood 2000 JAN
High levels of lymphoid expression of enhanced green fluorescent protein in nonhuman primates transplanted with cytokine-mobilized peripheral blood CD34(+) cells.
Abstract
Abstract
We have used a murine retrovirus vector containing an enhanced green fluorescent protein complimentary DNA (EGFP cDNA) to dynamically follow vector-expressing cells in the peripheral blood (PB) of transplanted rhesus macaques. Cytokine mobilized CD34(+) cells were transduced with an amphotropic vector that expressed EGFP and a dihydrofolate reductase cDNA under control of the murine stem cell virus promoter. The transduction protocol used the CH-296 recombinant human fibronectin fragment and relatively high concentrations of the flt-3 ligand and stem cell factor. Following transplantation of the transduced cells, up to 55% EGFP-expressing granulocytes were obtained in the peripheral circulation during the early posttransplant period. This level of myeloid marking, however, decreased to 0.1% or lower within 2 weeks. In contrast, EGFP expression in PB lymphocytes rose from 2%-5% shortly following transplantation to 10% or greater by week 5. After 10 weeks, the level of expression in PB lymphocytes continued to remain at 3%-5% as measured by both flow cytometry and Southern blot analysis, and EGFP expression was observed in CD4(+), CD8(+), CD20(+), and CD16/56(+) lymphocyte subsets. EGFP expression was only transiently detected in red blood cells and platelets soon after transplantation. Such sustained levels of lymphocyte marking may be therapeutic in a number of human gene therapy applications that require targeting of the lymphoid compartment. The transient appearance of EGFP(+) myeloid cells suggests that transduction of a lineage-restricted myeloid progenitor capable of short-term engraftment was obtained with this protocol. (Blood. 2000;95:445-452)