MethoCult™ H4431
Methylcellulose-based medium with agar-LCM and EPO for human cells
概要
MethoCult™ H4431 is a methylcelluose-based medium for the growth and enumeration of hematopoietic progenitor cells in colony-forming unit (CFU) assays of bone marrow, mobilized peripheral blood, peripheral blood, and cord blood samples. MethoCult™ H4431 supports the growth of erythroid progenitor cells (BFU-E and CFU-E), granulocyte-macrophage progenitor cells (CFU-GM, CFU-G and CFU-M), and multipotential granulocyte, erythroid, macrophage, megakaryocyte progenitor cells (CFU-GEMM). This formulation contains agar leukocyte conditioned medium (agar-LCM) as a source of colony-stimulating factors, plus recombinant human erythropoietin (EPO). It is suitable for use as a control medium for detection of “EPO-independent” erythroid progenitor cells using MethoCult™ H4531 (Catalog #04531).
Browse our Frequently Asked Questions (FAQs) on performing the CFU assay and explore its utility as part of the cell therapy workflow.
Browse our Frequently Asked Questions (FAQs) on performing the CFU assay and explore its utility as part of the cell therapy workflow.
Contains
• Methylcellulose in Iscove's MDM
• Fetal bovine serum
• Bovine serum albumin
• 2-Mercaptoethanol
• Agar-LCM
• Recombinant human erythropoietin (EPO)
• Supplements
• Fetal bovine serum
• Bovine serum albumin
• 2-Mercaptoethanol
• Agar-LCM
• Recombinant human erythropoietin (EPO)
• Supplements
Subtype
Semi-Solid Media, Specialized Media
Cell Type
Hematopoietic Stem and Progenitor Cells
Species
Human
Application
Cell Culture, Colony Assay, Functional Assay
Brand
MethoCult
Area of Interest
Stem Cell Biology
技术资料
| Document Type | 产品名称 | Catalog # | Lot # | 语言 |
|---|---|---|---|---|
| Product Information Sheet | MethoCult™ H4431 | 04431 | All | English |
| Manual | MethoCult™ H4431 | 04431 | All | English |
| Safety Data Sheet | MethoCult™ H4431 | 04431 | All | English |
数据及文献
Data
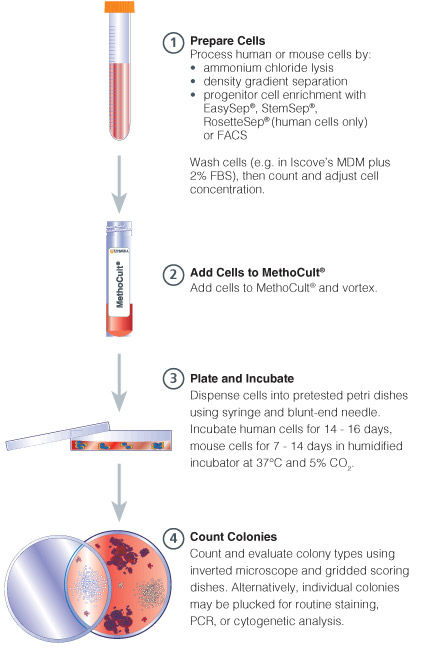
Figure 1. Procedure Summary for Hematopoietic CFU Assays
Publications (10)
Blood 2009 FEB
Clonal analysis of deletions on chromosome 20q and JAK2-V617F in MPD suggests that del20q acts independently and is not one of the predisposing mutations for JAK2-V617F.
Abstract
Abstract
We developed a real-time copy number polymerase chain reaction assay for deletions on chromosome 20q (del20q), screened peripheral blood granulocytes from 664 patients with myeloproliferative disorders, and identified 19 patients with del20q (2.9%), of which 14 (74%) were also positive for JAK2-V617F. To examine the temporal relationship between the occurrence of del20q and JAK2-V617F, we performed colony assays in methylcellulose, picked individual burst-forming units-erythroid (BFU-E) and colony-forming units-granulocyte (CFU-G) colonies, and genotyped each colony individually for del20q and JAK2-V617F. In 2 of 9 patients, we found that some colonies with del20q carried only wild-type JAK2, whereas other del20q colonies were JAK2-V617F positive, indicating that del20q occurred before the acquisition of JAK2-V617F. However, in colonies from 3 of 9 patients, we observed the opposite order of events. The lack of a strict temporal order of occurrence makes it doubtful that del20q represents a predisposing event for JAK2-V617F. In 2 patients with JAK2-V617F and 1 patient with MPL-W515L, microsatellite analysis revealed that del20q affected chromosomes of different parental origin and/or 9pLOH occurred at least twice. The fact that rare somatic events, such as del20q or 9pLOH, occurred more than once in subclones from the same patients suggests that the myeloproliferative disorder clone carries a predisposition to acquiring such genetic alterations.
Blood 2000 JAN
High levels of lymphoid expression of enhanced green fluorescent protein in nonhuman primates transplanted with cytokine-mobilized peripheral blood CD34(+) cells.
Abstract
Abstract
We have used a murine retrovirus vector containing an enhanced green fluorescent protein complimentary DNA (EGFP cDNA) to dynamically follow vector-expressing cells in the peripheral blood (PB) of transplanted rhesus macaques. Cytokine mobilized CD34(+) cells were transduced with an amphotropic vector that expressed EGFP and a dihydrofolate reductase cDNA under control of the murine stem cell virus promoter. The transduction protocol used the CH-296 recombinant human fibronectin fragment and relatively high concentrations of the flt-3 ligand and stem cell factor. Following transplantation of the transduced cells, up to 55% EGFP-expressing granulocytes were obtained in the peripheral circulation during the early posttransplant period. This level of myeloid marking, however, decreased to 0.1% or lower within 2 weeks. In contrast, EGFP expression in PB lymphocytes rose from 2%-5% shortly following transplantation to 10% or greater by week 5. After 10 weeks, the level of expression in PB lymphocytes continued to remain at 3%-5% as measured by both flow cytometry and Southern blot analysis, and EGFP expression was observed in CD4(+), CD8(+), CD20(+), and CD16/56(+) lymphocyte subsets. EGFP expression was only transiently detected in red blood cells and platelets soon after transplantation. Such sustained levels of lymphocyte marking may be therapeutic in a number of human gene therapy applications that require targeting of the lymphoid compartment. The transient appearance of EGFP(+) myeloid cells suggests that transduction of a lineage-restricted myeloid progenitor capable of short-term engraftment was obtained with this protocol. (Blood. 2000;95:445-452)
Blood 2000 JAN
Distinct signals control the hematopoiesis of lymphoid-related dendritic cells.
Abstract
Abstract
The molecular and cellular requirements for the development of different populations of human dendritic cells (DC) were studied. Conditions were defined that support DC production from lymphoid progenitors but that fail to induce DC formation from peripheral monocytes. The production of these lymphoid-related DC was severely blocked when hematopoietic progenitors overexpressed Ik7, a mutant dominant-negative Ikaros protein. In contrast, Ik7 did not block the formation of DC in conditions supporting the development of monocyte-derived DC. Furthermore, Ik7 did not block the formation of monocyte/macrophages and enhanced granulopoiesis. One of the molecular mechanisms mediated by Ik7 appears to be down-regulation of the flt3-receptor mRNA. Thus, distinct signals control the formation of DC demonstrating that some aspects of DC diversity are determined in part by distinct molecular cues at the hematopoietic level. (Blood. 2000;95:128-137)
Experimental hematology 1999 NOV
Comparison of in vitro drug-sensitivity of human granulocyte-macrophage progenitors from two different origins: umbilical cord blood and bone marrow.
Abstract
Abstract
Predictive in vitro hematotoxicity assays using human cells will provide estimation of tolerable level and aid considerably the development of agents with greater therapeutic activity and less toxicity. Human hematopoietic cells can be derived from three sources: human bone marrow by sternal or femoral aspiration, mobilized peripheral blood, or umbilical cord blood samples collected from placentas after deliveries. Because of the difficulties to have a continuous supply of bone marrow cells from normal human donors and the related ethical problems, we performed a study to compare the sensitivity of human bone marrow cells (h-BMC) and human cord blood cells (h-CBC) to chemicals in order to confirm if h-CBC can readily replace bone marrow cells in checking the sensitivity of GM-CFU progenitors to drugs as preliminarily reported in literature. Our results showed that the prediction of IC50 values in human model is quite similar by using h-BMC or h-CBC. On the contrary, the type of medium influenced in a significant way the ICs determination of some drugs.
Experimental hematology 1999 AUG
Bispecific antibody fragments with CD20 X CD28 specificity allow effective autologous and allogeneic T-cell activation against malignant cells in peripheral blood and bone marrow cultures from patients with B-cell lineage leukemia and lymphoma.
Abstract
Abstract
Bispecific antibodies directed against tumor-associated target antigens and to surface receptors mediating T-cell activation, such as the TCR/CD3 complex and the costimulatory receptor CD28, are capable of mediating T-cell activation resulting in tumor cell killing. In this study, we used the B-cell-associated antigens CD19 and CD20 as target structures on human leukemic cells. We found that a combination of bispecific antibody fragments (bsFab2) with target x CD3 and target x CD28 specificity induces vigorous autologous T-cell activation and killing of malignant cells in peripheral blood and bone marrow cultures from patients with chronic lymphocytic leukemia and follicular lymphoma. The bsFab2 targeting CD20 were considerably more effective than those binding to CD19. The colony-forming capacity of treated bone marrow was impaired due to large amounts of tumor necrosis factor alpha produced during bsFab2-induced T-cell activation. Neutralizing tumor necrosis factor alpha antibodies were found to reverse this negative effect without affecting T-cell activation and tumor cell killing. CD20 x CD28 bsFab2, when used alone rather than in combination, markedly improved the recognition of leukemic cells by allogeneic T cells. Therefore, these reagents may be capable of enhancing the immunogenicity of leukemic cells in general and, in particular, of increasing the antileukemic activity of allogeneic donor buffy coat cells in relapsed bone marrow transplanted patients.
Experimental hematology 1999 AUG
Retroviral vector-mediated gene transfer into umbilical cord blood CD34brCD38-CD33- cells.
Abstract
Abstract
In this report, we sought to optimize gene transfer into primitive human umbilical cord blood (UCB) cells. Initially, we found that fresh UCB isolated with the CD34brCD38 CD33 phenotype were highly enriched for hematopoietic progenitors detected in extended long-term cultures (8-week LTCs). In addition, following ex vivo gene transfer, this population possessed virtually all the 8-week LTC activity of the cultured cells. A multiparameter FACS assay was developed to efficiently screen the effects of alternative retroviral vector gene transfer procedures on the transduction efficiency and maintenance of CD34brCD38 CD33 cells. Proliferation of the CD34brCD38 CD33 cells was found to be a prerequisite for efficient transduction. However, in all conditions tested, proliferation of the CD34brCD38 CD33 cells was associated with a progressive loss of primitive cell properties including a reduction in CD34 expression, an increase in CD38/CD33 expression, and a decline in the ability to sustain 8-week LTCs. These observations indicate that it will be necessary to define conditions that more effectively support the self-renewal capacity of CD34brCD38 CD33 cells to optimize retroviral vector gene transfer in these cells. Evaluating these conditions and reagents will be facilitated by the multiparameter FACS assay described in this report.