STEMdiff™ Forebrain Neuron Differentiation Kit
• Supports highly efficient generation of functional neurons from hPSC-derived neuronal precursors
• Produces a highly pure population (≥ 90% neurons) of mixed excitatory and inhibitory neurons that can be maintained long-term in culture
• Optimized for differentiation of neuronal progenitor cells generated using STEMdiff™ SMADi Neural Induction Kit
• Supports neuronal activity for physiologically relevant results
• Enables reproducible maturation of neuronal precursors derived from multiple human ES and iPS cell lines
- STEMdiff™ Forebrain Neuron Differentiation Basal Medium, 80 mL
- STEMdiff™ Forebrain Neuron Differentiation Supplement, 20 mL
| Document Type | 产品名称 | Catalog # | Lot # | 语言 |
|---|---|---|---|---|
| Product Information Sheet | STEMdiff™ Forebrain Neuron Differentiation Kit | 08600 | All | English |
| Special Protocol | STEMdiff™ Forebrain Neuron Differentiation Kit | 08600 | All | English |
| Safety Data Sheet 1 | STEMdiff™ Forebrain Neuron Differentiation Kit | 08600 | All | English |
| Safety Data Sheet 2 | STEMdiff™ Forebrain Neuron Differentiation Kit | 08600 | All | English |
Data
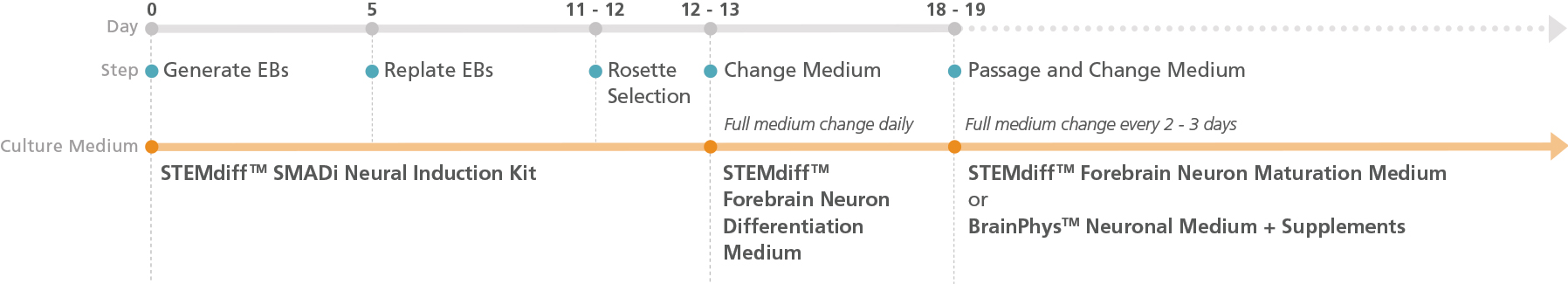
Figure 1. Schematic for the Embryoid Body Protocol
Forebrain-type neural precursors can be generated in 18 - 19 days from hPSC-derived neural progenitor cells (NPCs) after selecting neural rosettes from replated embryoid bodies. For the maturation of precursors to forebrain-type neurons, see the PIS.

Figure 2. Schematic for the Monolayer Protocol
Forebrain-type neural precursors can be generated from neural progenitor cell (NPC) monolayers derived from embryonic and induced pluripotent stem cells after three single-cell passages. For the maturation of precursors to forebrain-type neurons, see the PIS.
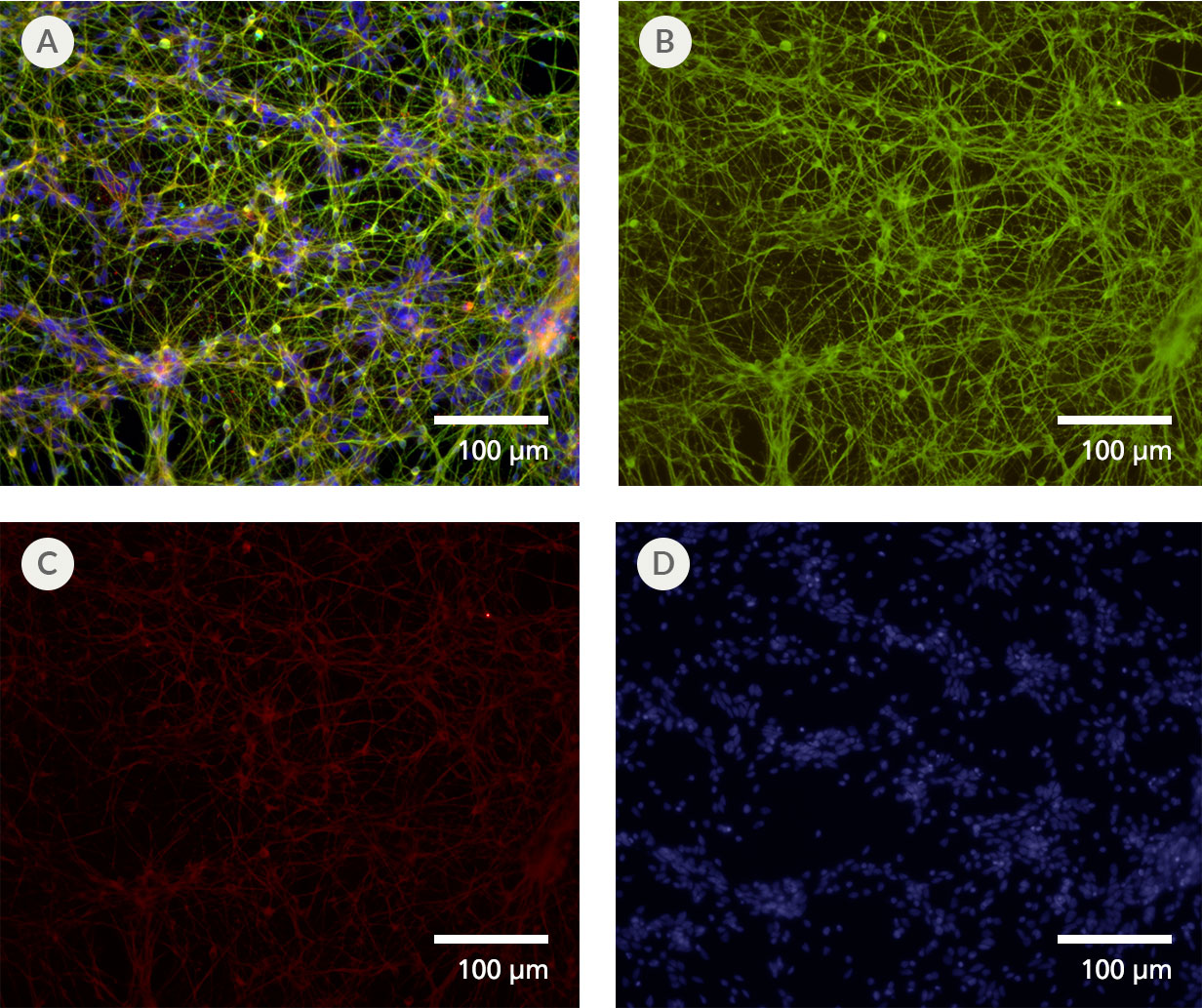
Figure 3. Forebrain-Type Neurons Are Generated After Culture in STEMdiff™ Forebrain Neuron Differentiation and Maturation Kits
NPCs generated from hPSCs in mTeSR 1™ using the STEMdiff™ SMADi Neural Induction Kit embryoid body (EB) protocol were differentiated and matured to forebrain-type neurons using the STEMdiff™ Forebrain Neuron Differentiation and Maturation Kits. (A) Forebrain-type neurons were formed after iPS cell-derived NPCs were cultured with the STEMdiff™ Forebrain Neuron Differentiation Kit for 7 days and STEMdiff™ Forebrain Neuron Maturation Kit for 14 days. The resulting cultures contain a highly pure population of (B) class III β-tubulin-positive neurons (green), with (C) fewer than 10% astrocytes (GFAP-positive cells, red). (D) Nuclei are labeled with DAPI (blue).
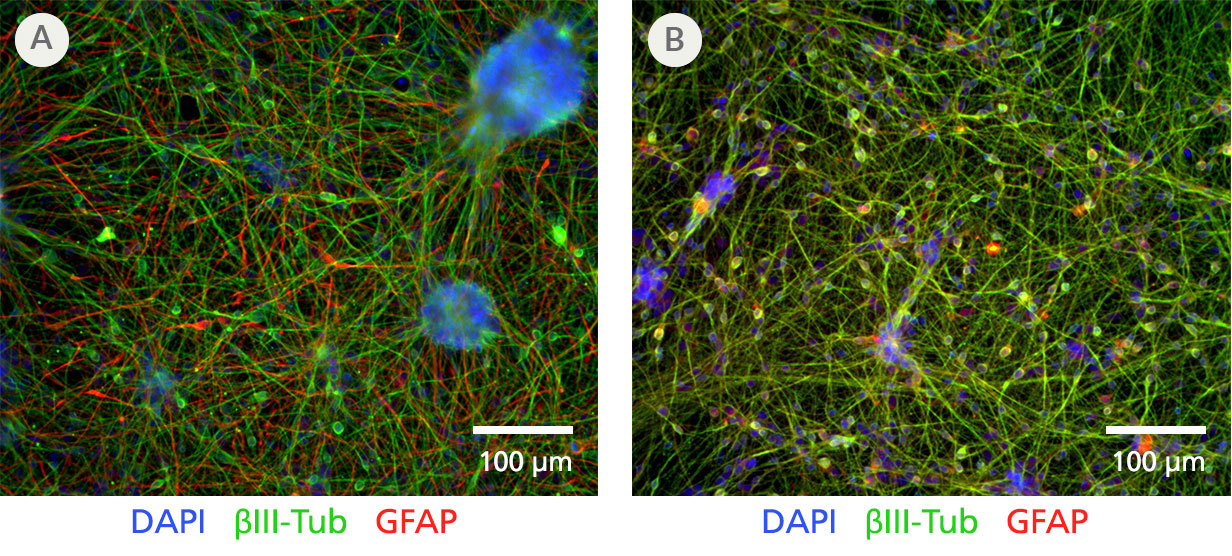
Figure 4. Performance of STEMdiff™ Neuron and the STEMdiff™ Forebrain Neuron Differentiation and Maturation Kits
Neural progenitor cells (NPCs) were derived using the monolayer protocol for the STEMdiff™ SMADi Neural Induction Kit, from H9 ES cells maintained in mTeSR™ 1. NPCs were then differentiated into forebrain-type neurons using either A) STEMdiff™ Neuron Differentiation and Maturation Kits or B) the equivalent-performing STEMdiff™ Forebrain Neuron Differentiation and Maturation Kits according to protocol instructions (see product information sheets for Catalog #08600/08605). The resulting neurons were fixed for immunofluorescence. Both sets of products demonstrate equivalent differentiation efficiency, with a yield of ≥90% class III ꞵ-tubulin-positive neurons (green) and ≤10% GFAP-positive astrocytes (red).
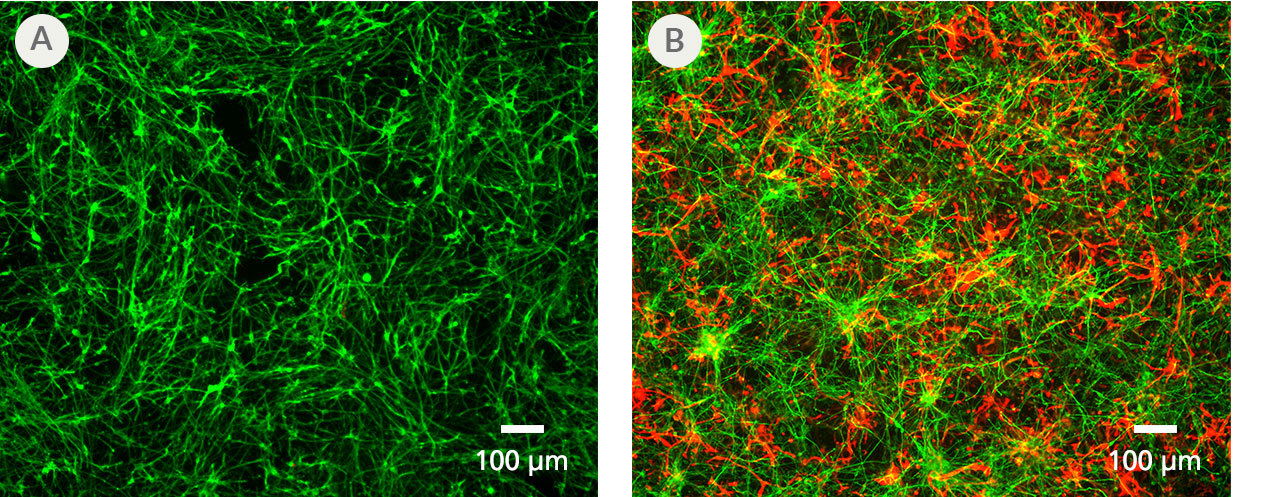
Figure 5. PSC-Derived Astrocytes and Neurons Can Be Co-Cultured to Model Cell-Cell Interactions In Vitro
NPCs generated from the H1 cell line were differentiated to astrocytes using STEMdiff™ Astrocyte Differentiation and Maturation Kits. H9 cell-derived NPCs were differentiated to forebrain-type neurons using STEMdiff™ Forebrain Neuron Differentiation and Maturation Kits. For co-culture, matured astrocytes were seeded onto forebrain neurons that had been in STEMdiff™ Forebrain Neuron Maturation Medium for at least one week. Co-cultures were then switched to STEMdiff™ Forebrain Neuron Maturation Medium the following day and for the remaining co-culture. (A) Neurons cultured alone, following the co-culture feeding schedule, are labeled with DCX (green). (B) DCX-positive neurons (green) and astrocytes (GFAP, red) can be co-cultured for at least 1 - 2 weeks prior to analysis. For a detailed co-culture protocol, please see the Methods Library.
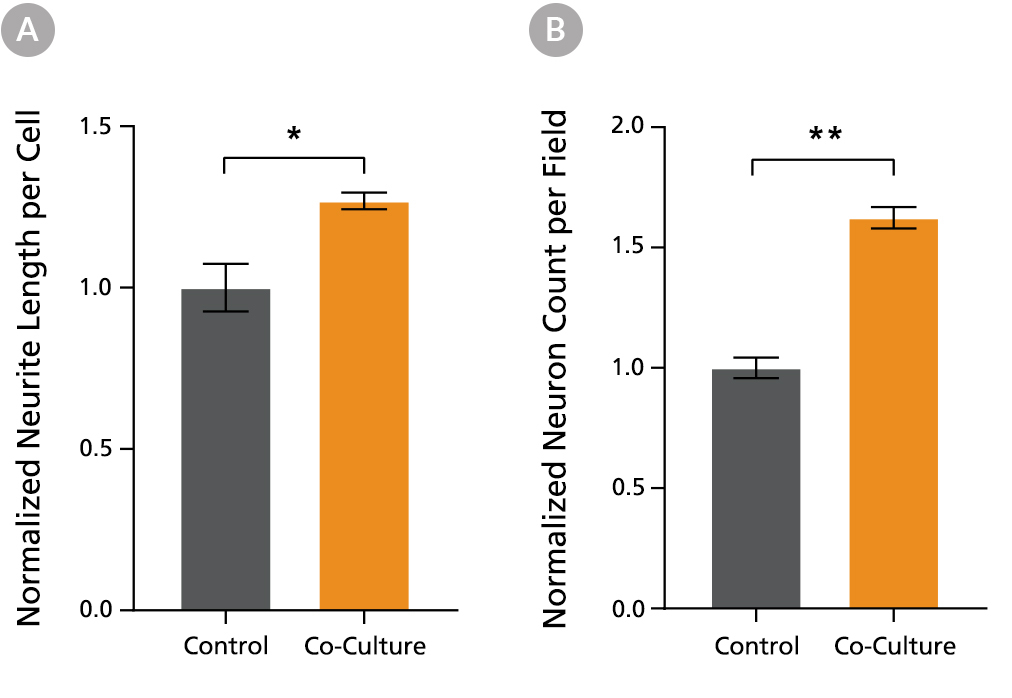
Figure 6. PSC-Derived Neurons Survive and Mature when Co-Cultured with PSC-Derived Astrocytes
NPCs generated from the STiPS-R038 cell line were differentiated to astrocytes using STEMdiff™ Astrocyte Differentiation and Maturation Kits. STiPS-M001 cell-derived NPCs were differentiated to forebrain-type neurons using STEMdiff™ Forebrain Neuron Differentiation and Maturation Kits. After co-culture for one week, neurons (A) had significantly increased neurite outgrowth as measured on MAP2-positive neurons with the NeuriteTracer plugin for ImageJ (M Pool et al. J Neurosci Methods, 2008) and (B) were more numerous than neurons cultured alone using the same feeding schedule. *, p < 0.05; **, p < 0.01.